Phe-136′ forms hydrophobic contacts with Ile-51, Lys-57, Met-59, and Leu-61 in the cAMP-bound state but not in the cAMP-free state. ( D) View from above the C-helices highlighting crucial contacts made by residues located at the C-terminal of the C-helices. ( C) View from above DNA showing the rigid-body rotation (indicated on the left subunit) and translation (indicated on the right subunit) that DBD undergoes upon cAMP binding. In apo-CAP, the C-helix extends only to Gln-125 in contrast, in CAP-cAMP 2-DNA (and CAP-cAMP 2) the C-helix extends to Phe-136. ( B) Close-up view of the intersubunit C-helix/C′helix coiled coil. Proteins are shown as cartoons and DNA as sticks in semitransparent surface. DNA is in light gray, and cAMP in dark gray. ( A) Superposition of apo-CAP (orange) and CAP-cAMP 2-DNA (blue) on the CBD (residues 10–125). Dotted lines denote polar contacts (hydrogen bonds or salt bridges). Structural comparison of apo-CAP and CAP-cAMP 2-DNA. 
The results define the structural mechanisms that underlie allosteric control of this prototypic transcriptional regulatory factor and provide an illustrative example of how effector-mediated structural changes can control the activity of regulatory proteins. The allosteric transition is stabilized further by expulsion of an aromatic residue from the cAMP-binding pocket upon cAMP binding. Binding of cAMP results in a coil-to-helix transition that extends the coiled-coil dimerization interface of CAP by 3 turns of helix and concomitantly causes rotation, by approximately 60 degrees, and translation, by approximately 7 A, of the DNA-binding domains (DBDs) of CAP, positioning the recognition helices in the DBDs in the correct orientation to interact with DNA. The structural changes, and their relationship to cAMP binding and DNA binding, are remarkably clear and simple. Here we report the structure of CAP in the absence of cAMP, which, together with structures of CAP in the presence of cAMP, defines atomic details of the cAMP-mediated allosteric transition. At another level, the role of cyclic AMP is more obvious: insulin deficiency leaves unopposed the actions of hormones which stimulate the production of cyclic AMP, thereby contributing to the glucose plethora and ketosis so often seen in the later stages of the disease.The cAMP-mediated allosteric transition in the catabolite activator protein (CAP also known as the cAMP receptor protein, CRP) is a textbook example of modulation of DNA-binding activity by small-molecule binding. This could be secondary to basement membrane thickening, but there is also evidence that the cyclic AMP mechanism may be defective. Whether or not cyclic AMP plays a regulatory role in basement membrane synthesis is presently unknown.Īnother defect recognizable in prediabetics is faulty insulin release in response to glucose infusion. Further study of the formation and breakdown of the basement membrane may therefore lead to a better understanding of the genetic defect. One line of evidence implicates basement membrane thickening as an early event in the patho genesis of diabetes. Human diabetes mellitus is recognized as the result of a basic genetic defect, the nature of which is undefined. Since cyclic AMP is involved in the release as well as several of the actions of insulin, the possible role of cyclic AMP in diabetes has been discussed. Cyclic AMP is thus seen to mediate the actions of several catabolic hormones as well as promote the release of an anabolic hormone which acts in part by opposing cyclic AMP. Insulin then travels to the liver and adipose tissue to suppress the accumulation of cyclic AMP, and may also antagonize the action of cyclic AMP in muscle. 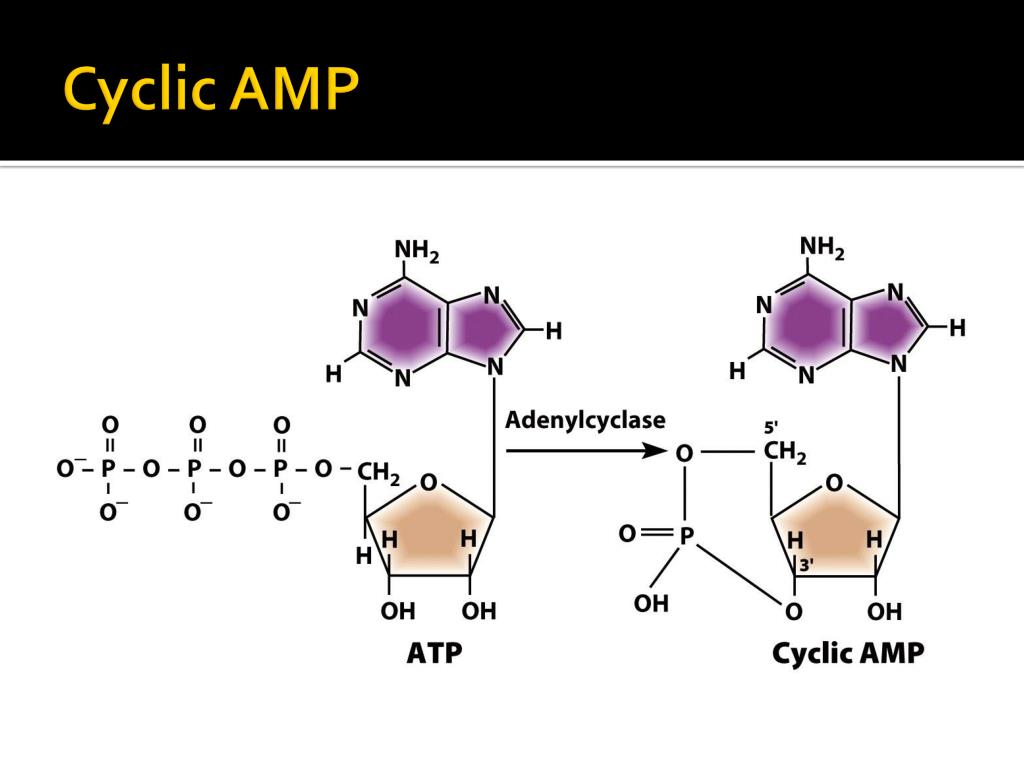
Among the principal effects of cyclic AMP in these tissues are glycogenolysis in muscle and lipolysis in adipose tissue.Īnother role of cyclic AMP is to enhance or promote the release of insulin from pancreatic beta cells. 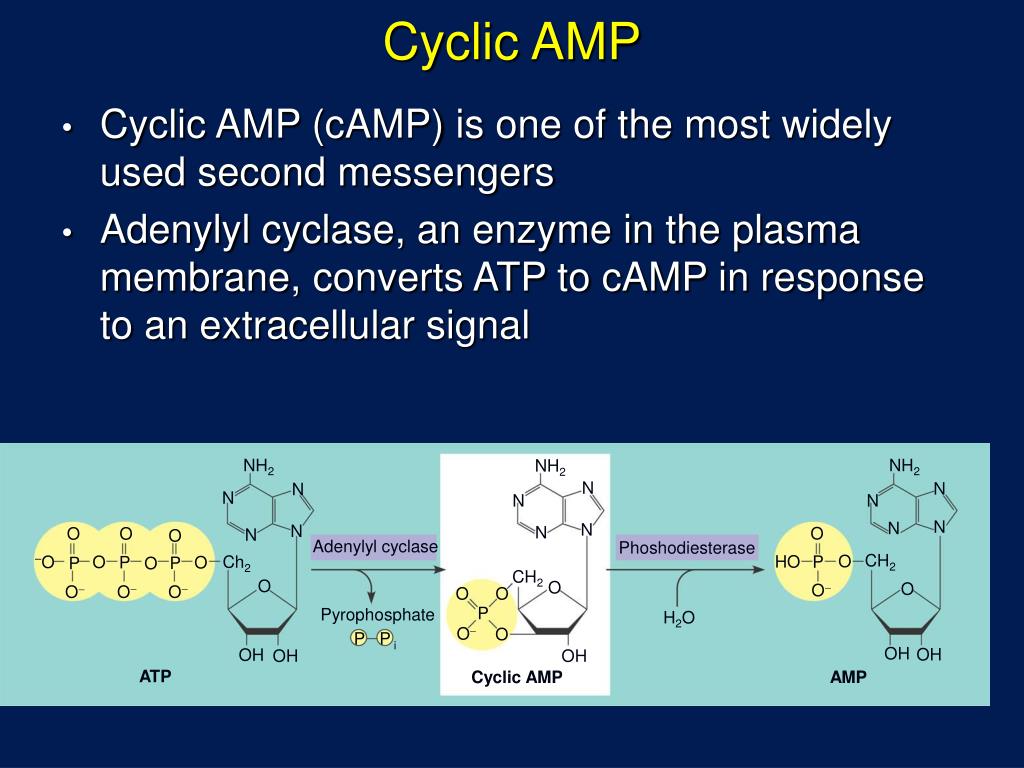
The catecholamines also stimulate adenyl cyclase in muscle and adipose tissue. cyclic AMP leads to a net increase in hepatic glucose production by at least three mechanisms: stimulation of phosphorylase activation, suppression of glycogen synthetase activity, and stimulation of gluconeogenesis. In the liver, glucagon and the catecholamines cause an increase in the intracellular level of cyclic AMPby stimulating adenyl cyclase. The chief role of cyclic AMP in several tissues seems to be to facilitate or promote the mobilization of glucose and fatty acid reserves. Emphasis in the present review has been placed on carbohydrate metabolism, but lipid metabolism has also been discussed to some extent. Cyclic AMP plays an important role in the regulation of metabolism generally.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
